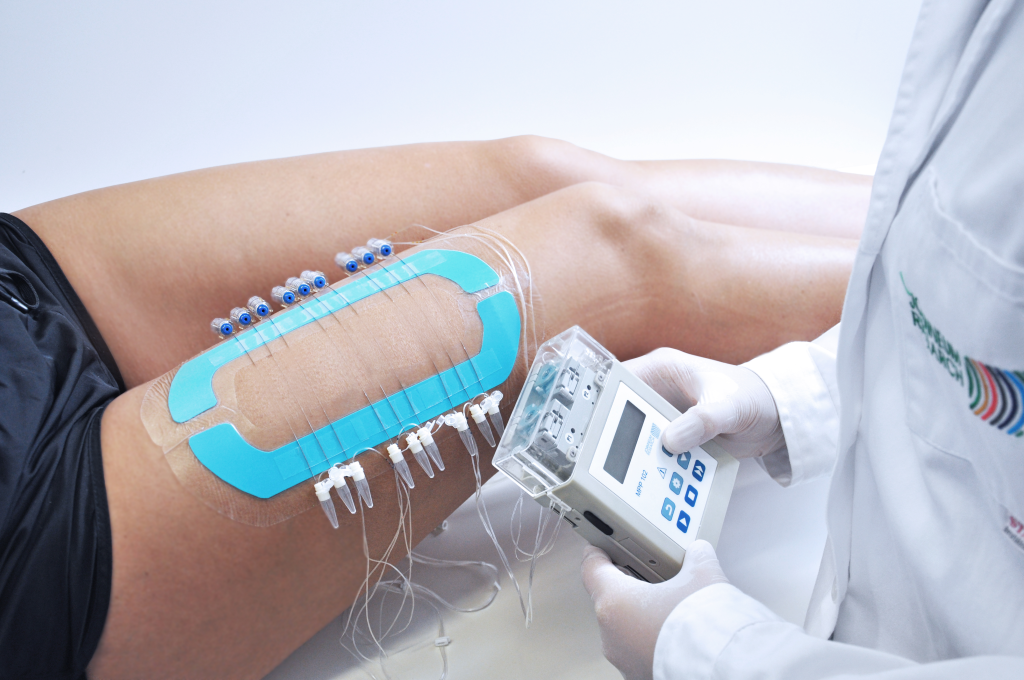
Open Flow Microperfusion
OFM is a minimally-invasive interface to peripheral tissues for continuous sampling of interstitial fluid in pharmacokinetic and pharmacodynamic studies.
Products
Different OFM products are specifically designed to optimize sampling in various tissues.
OFM working principle
Technology
OFM is a membrane-free, time-resolved, minimally-invasive, in-vivo sampling technology developed by JOANNEUM RESEARCH.
OFM uses probes with macroscopic openings to exchange substances in a liquid pathway, that is based on concection and diffusion, so that the exchange can occur non-selectively in either direction.
Project Partners





Your benefits
Universal
OFM samples all substances from the interstitial fluid independent of size, lipophilicity or protein-binding.
Targeted
OFM probes enable continuous sampling from clearly specified tissue types.
Cost and time efficient
OFM studies reduce costs and time by providing a complete pharmacological profile early in drug development.
Medical Device Regulation
Open Flow Microperfusion (OFM) is a CE-marked medical device technology for the minimally-invasive collection of analytes from the interstitial fluid of defined tissues. The OFM system enables continuous sampling in dermal tissue and subcutaneous adipose tissue in adult subjects within approved clinical studies.
Any additional research applications described on this website refer to the OFM methodology and are not part of the intended use of the CE-marked medical device. Applications outside the intended use require separate regulatory evaluation and approval.