- About us
- Research fields
- DIGITAL – Institut für Digitale Technologien
- MATERIALS – Institut für Sensorik, Photonik und Fertigungstechnologien
- ROBOTICS – Institut für Robotik und Flexible Produktion
- COREMED – Zentrum für Regenerative Medizin und Präzisionsmedizin
- HEALTH – Institut für Biomedizinische Forschung und Technologien
- LIFE – Institut für Klima, Energiesysteme und Gesellschaft
- POLICIES – Institut für Wirtschafts-, Sozial und Innovationsforschung
- Business areas
- Products & Services
- Research infrastructure
- Beteiligungen
- Career
- Aktuelles
- Publications
- Kontakt zu uns
Dermal Bioequivalence Studies

Fast and efficient PK-based bioequivalence studies for topical generics

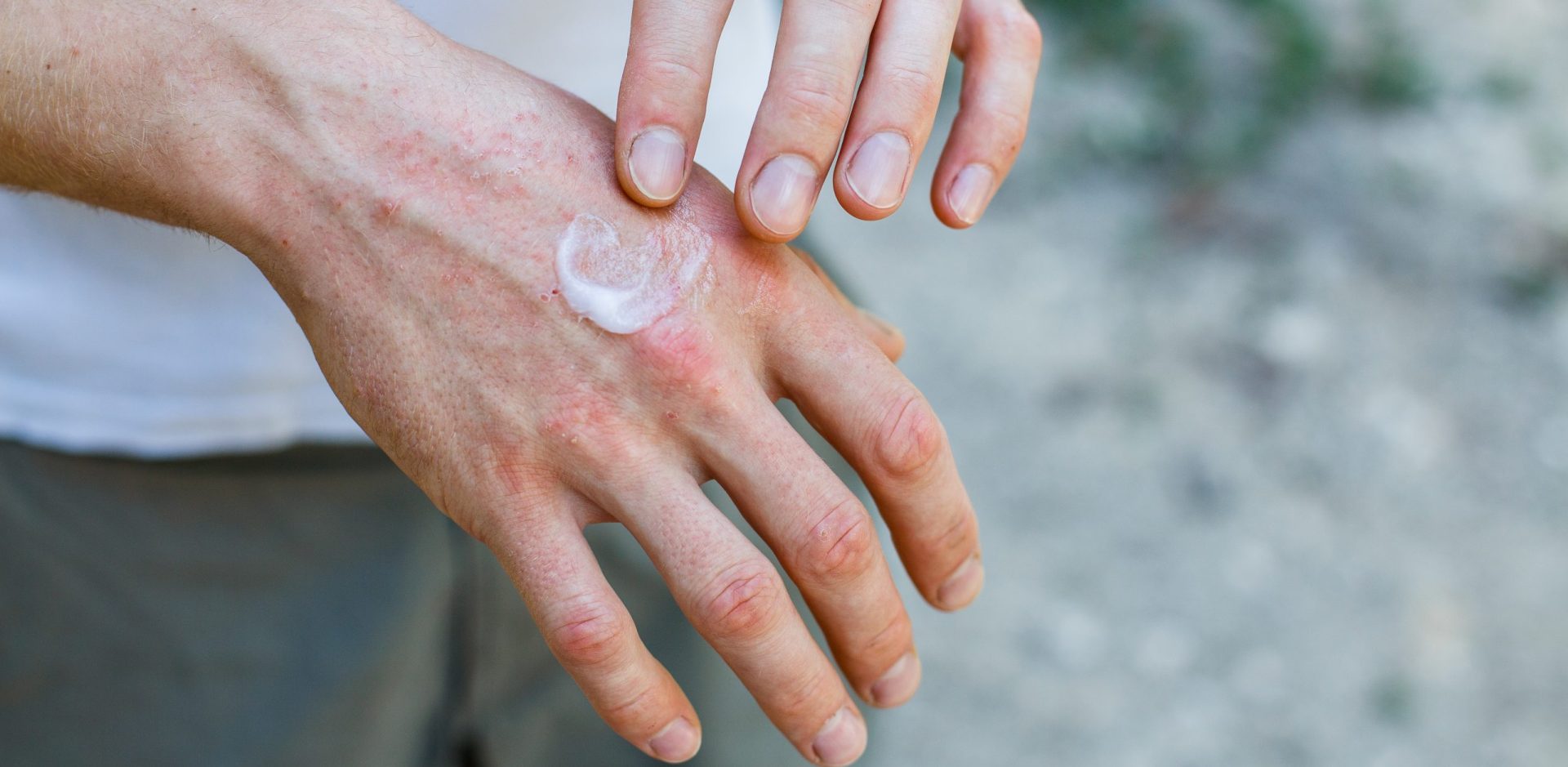
Our innovative dOFM Technology provides dermal bioequivalence (BE) data using a pharmacokinetic-based approach. Currently, FDA approval for topical generics requires a clinical endpoint study to demonstrate bioequivalence. Standard PK studies with blood samples are not suitable for BE testing of topical dermal generics. dOFM enables a PK-based BE approach directly in the skin.
Our Services
- PK-based approach for bioequivalence tests directly in the skin
- Test and reference drug are compared at the same time on the same subject
- Strategic advice for product-specific guidelines and ANDA meetings (505b2, product development meeting)
- All services required for an FDA submission are provided by our team and our partners
Your Benefits
- one PK study instead of an expensive clinical endpoint study
- fewer participants (< 50 healthy volunteers) instead of hundreds of patients in a BE study
- a single center instead of a multicenter study
- our approach has a lower risk of failure compared to clinical endpoint studies
You are currently viewing a placeholder content from Youtube. To access the actual content, click the button below. Please note that doing so will share data with third-party providers.
More Information
Research groups
Downloads
Unsere beteiligten Partner & Fördergeber
Das sagen unsere Kunden
Publikationen zum Produkt oder Service
More Products and Services
We handle the full validation lifecycle for REDCap — so your clinical trial runs on a system regulators trust and your team can rely on.
aOFM can be used to monitor local drug distribution and biomarkers in subcutaneous adipose tissue.
With cOFM, local drug distribution and biomarkers in the brain can be monitored - with the blood-brain barrier intact.
Unique de-risking and proof-of-mechanism studies for dermal active ingredients
Specialized methods for analyses and testing for preclincial and clinical studies (GLP and non-GLP).
Instruments and methods for the measurement of pharmacokinetics and pharmacodynamics in different tissues.
Comprehensive and compliant data management and biostatistics for clinical trials
We test drug release in a fast and reliable way using IVRT based on vertical diffusion cells and synthetic membranes.
We perform untargeted and targeted metabolomics studies to quantify metabolites to investigate metabolic processes and identify biomarkers.
The digital checklist and cross-professional process documentation for surgical procedures.
Unlock success in regulatory compliance for medical devices with tailored, hands-on guidance
Miteinander zukunftsrelevant
Die JOANNEUM RESEARCH ist Innovations- und Technologieanbieter im Bereich der angewandten Forschung. Als Forschungsgesellschaft der Länder und Regionen prägen wir mit unseren Forschungskompetenzen die Entwicklung unserer modernen Gesellschaft und Wirtschaft nachhaltig und menschenzentriert. Als multidisziplinäres Team in flexiblen, innovationsfreundlichen Strukturen leben wir höchste gesellschaftliche und wissenschaftliche Ansprüche.












