- About us
- Research fields
- DIGITAL – Institut für Digitale Technologien
- MATERIALS – Institut für Sensorik, Photonik und Fertigungstechnologien
- ROBOTICS – Institut für Robotik und Flexible Produktion
- COREMED – Zentrum für Regenerative Medizin und Präzisionsmedizin
- HEALTH – Institut für Biomedizinische Forschung und Technologien
- LIFE – Institut für Klima, Energiesysteme und Gesellschaft
- POLICIES – Institut für Wirtschafts-, Sozial und Innovationsforschung
- Business areas
- Products & Services
- Research infrastructure
- Beteiligungen
- Career
- Aktuelles
- Publications
- Kontakt zu uns
Pharmacological testing of subcutaneous drugs

aOFM can be used to monitor local drug distribution and biomarkers in subcutaneous adipose tissue.

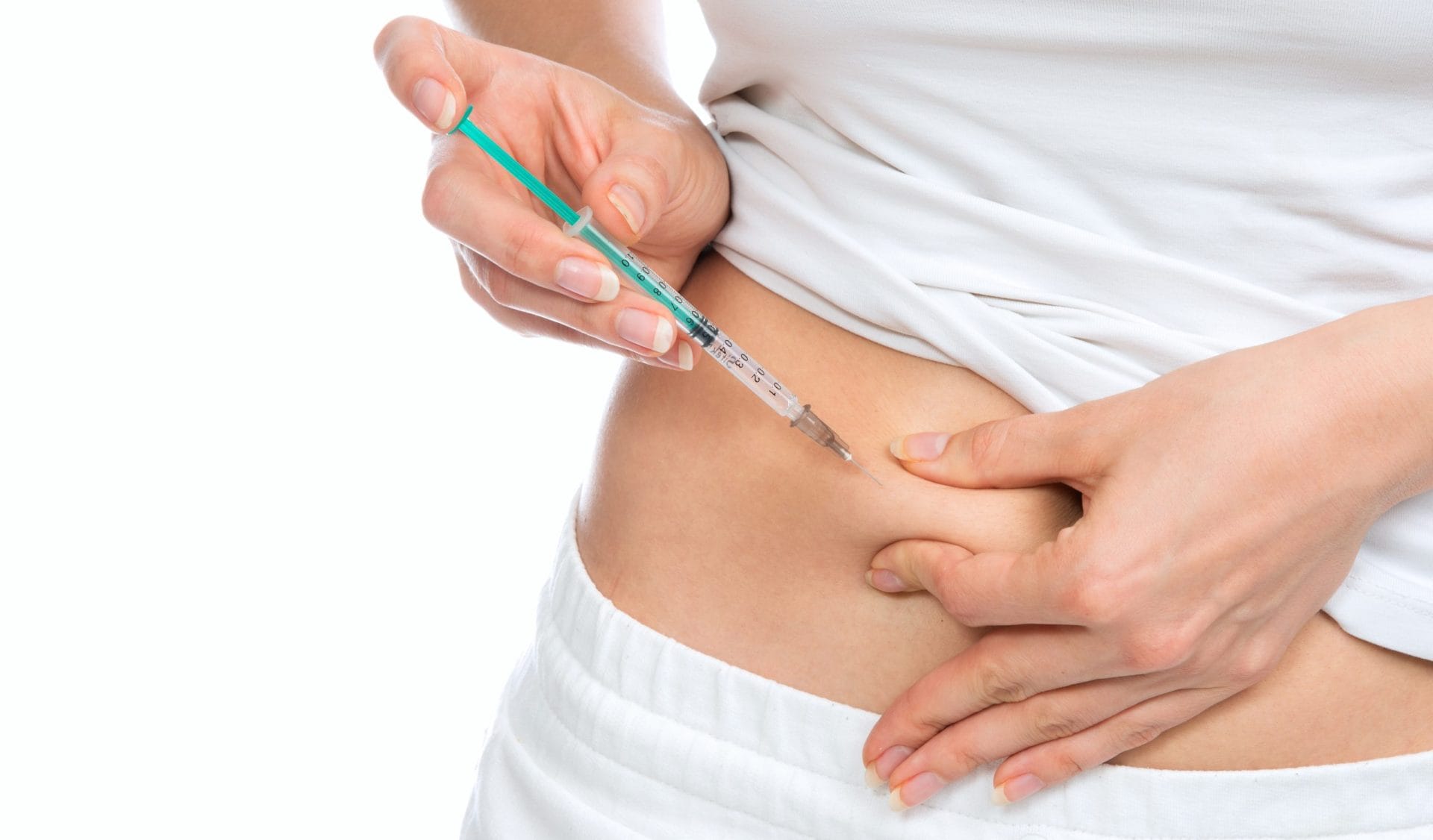
Injection into the subcutaneous fatty tissue is an important route of administration for many substances such as insulin or antibodies. The formation of drug depots in adipose tissue and drug distribution are crucial for assessing absorption into the blood, pharmaceutical effects or possible side effects. aOFM studies provide data on substance distribution in adipose tissue and pharmacodynamic effects in the target tissue.
Preclinical studies
Ex vivo experiments are carried out with freshly removed human and animal tissues. Our studies include animal models in awake and anaesthetized animals (rodents, pigs).
Clinical studies
Clinical trials range from first-in-human studies to phase 0 or phase 1 clinical trials in which subcutaneously administered drugs can be monitored for up to 36 hours using aOFM or microdialysis. Clinical trials can be conducted in healthy volunteers or in patients (diabetes type 1 and 2). Drug concentrations and PD markers are analyzed in our GLP-certified laboratory.
Several samples per application enable an efficient studythat creates a large set of reliable data, while only a small number of participants are needed for PK/PD studies.
Clinical trials are conducted according to ICH-GCP standards at the Clinical Trials Unit of the Medical University of Graz.
Additional expertise
- Combination of microdialysis and OFM technologies for protein binding studies.
- Testing and optimization of injection devices: Use of live in vivo imaging techniques (e.g., µCT) to visualize an injected substance depot directly in the tissue.
- Various glucose clamp techniques (hypo-, hyper-, euglycemia) for determining the glucose infusion rate, insulin initiation time, etc.
- Studies with isotope-labeled insulins for monitoring glucose metabolism (endogenous glucose production and peripheral glucose uptake)
Research groups
Downloads
Unsere beteiligten Partner & Fördergeber
Das sagen unsere Kunden
Publikationen zum Produkt oder Service
More Products and Services
We handle the full validation lifecycle for REDCap — so your clinical trial runs on a system regulators trust and your team can rely on.
Fast and efficient PK-based bioequivalence studies for topical generics
With cOFM, local drug distribution and biomarkers in the brain can be monitored - with the blood-brain barrier intact.
Unique de-risking and proof-of-mechanism studies for dermal active ingredients
Specialized methods for analyses and testing for preclincial and clinical studies (GLP and non-GLP).
Instruments and methods for the measurement of pharmacokinetics and pharmacodynamics in different tissues.
Comprehensive and compliant data management and biostatistics for clinical trials
We test drug release in a fast and reliable way using IVRT based on vertical diffusion cells and synthetic membranes.
We perform untargeted and targeted metabolomics studies to quantify metabolites to investigate metabolic processes and identify biomarkers.
The digital checklist and cross-professional process documentation for surgical procedures.
Prozessoptimierung in der Akutgeriatrie
Miteinander zukunftsrelevant
Die JOANNEUM RESEARCH ist Innovations- und Technologieanbieter im Bereich der angewandten Forschung. Als Forschungsgesellschaft der Länder und Regionen prägen wir mit unseren Forschungskompetenzen die Entwicklung unserer modernen Gesellschaft und Wirtschaft nachhaltig und menschenzentriert. Als multidisziplinäres Team in flexiblen, innovationsfreundlichen Strukturen leben wir höchste gesellschaftliche und wissenschaftliche Ansprüche.











